Imagine if you could take a blood test that would help you to detect 50 different cancers earlier than normal and receive lifesaving treatment. This testing is called a “liquid biopsy” (aka a normal blood draw) and early results are promising. I want to explore this topic with you in a little greater depth.
Cancer Screening
Second only to heart disease, cancer is the leading cause of death in people under 80. One in every four of us will die of cancer. Survival rates for certain cancers have improved dramatically over recent years; however, outcomes remain poor for others like pancreatic and lung cancer. Early diagnosis allows for less invasive and potentially curative treatment and typically better outcomes in most cancer treatments.
For many years, the research community has sought to discover the “holy grail” in cancer screening – a single reliable blood test that can detect multiple cancers in their early stages. Until recently, screening tests were for a few specific cancers (like lung, breast, colorectal, cervical, and prostate). In the last five years a handful of liquid biopsy tests have emerged.
What is a liquid biopsy?
First, it’s a normal blood draw, no different than drawing your blood like you do normally at the doctor’s office. The term “liquid biopsy” relates to measuring tumor cells or biomarkers (nucleic acids) circulating in the blood, with a specificity close to that of a tissue biopsy. This approach allows us a minimally invasive approach to identify, monitor, and treat disease. The traditional tissue biopsy involves the biopsy of suspected tumor tissue, a procedure that is invasive and with risks. Liquid biopsy is a screening test and not a diagnostic test, unlike tissue biopsy. There is an abundance of work being done in Europe and the US.1 Some liquid Biopsies exist and are FDA approved for monitoring of existing cancers and their treatment, including CELLSEARCH, FoundationOne Liquid CDx and Guardant360 CDx.
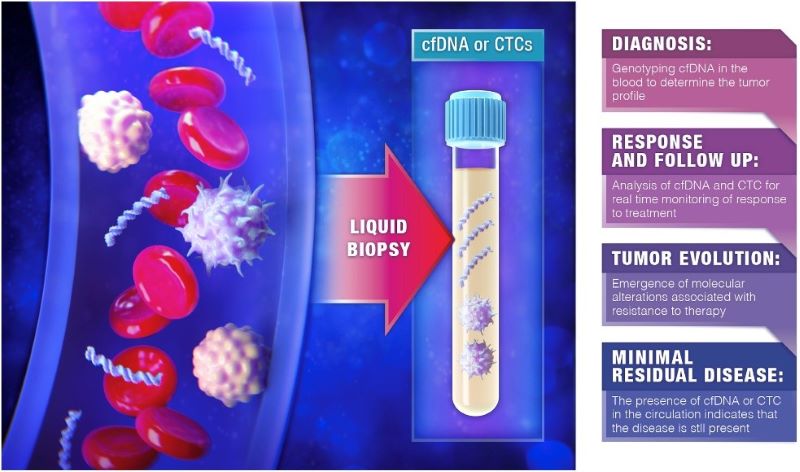
Promising Tests
Over the past few years research has identified several highly promising tests. The CancerSEEK assay, Galleri test, and the PanSeer assays are in clinical trials.
CancerSEEK assay has the potential to identify 8 or more cancers: ovarian, liver, stomach, pancreatic, esophageal, colorectal, breast, and lung. The specificity of the test was over 99% in eight cancer types, meaning that in 99% of cases with a positive test cancer was detected. The reliability depended on the type of cancer and the staging, early-stage cancers were often missed.
The most exciting is the Galleri test (developed by GRAIL) which aims to detect 50 types of cancer, for which ONLY five have screening tests. The Galleri test is currently the test under the most clinical trials in the UK and US.
A larger sub-study conducted of 6,689 participants (2,482 with cancer) reported 67.3% sensitivity (positive test and positive for cancer) for stages I-III (pre-metastatic) for 12 common cancers and 43.9% for all cancer types.2
Early results of the US-based PATHFINDER multi-center study had a 55% sensitivity across, a false positive rate of <1%. The sensitivity appears to be highly variable depending on the type of cancer and staging. For instance, Galleri has been shown to detect just 18.2% of kidney cancers compared to 93.5% of lung cancers. While overall it picks up 90.1% of stage IV cancers, it only detects an average of 16.8% of stage I cancers.3
This research, while promising, does not satisfy the high standard expected in clinical research: they are promoting other smaller studies prior to the results of the large PATHFINDER study conclusion. Large trials are underway including PATHFINDER in the USA and another (165,000 people) in the UK under the National Health Service (NHS)—results will not be available for some time.4
Despite the lack of validated research at this stage and FDA approval, the Galleri test is becoming increasingly available in the US, being promoted and marketed directly to consumers, primary care, and concierge physicians.5 It is not currently recommended as a screening test by the American Cancer Society.
While this tactic may be a smart business move, from a beneficence and non-maleficence point of view, there’s plenty of risk.
Potential Problems with liquid biopsy
It’s too early. The companies and the media are promoting tests before adequate research demonstrates efficacy.
Collection methods. Researchers are still trying to determine the volume of blood sample needed. In early stage, cancers there are very few circulating cancer cells, so large volumes of blood would be needed to detect cells.6
False negatives. A negative test is cause for celebration. However, sometimes screening tests miss cancer (false negative), reducing other screening tests, lulling clinician and patient into a false sense of security (and potentially leading to a missed diagnosis of cancer). Moreover, patients may be more inclined to skip some of their regular screening tests for cancer (e.g., Colonoscopy, Pap, Mammo, etc.), placing too much emphasis on the accuracy of the results of such tests.7,8
False positives. A positive test in a patient with no cancer can create unnecessary fear, leading to invasive testing and even increased risk of depression. These seem to be low in early testing.
Overtreatment. Some types of cancer are extremely slow growing (like prostate cancer in older men). An earlier diagnosis does not always necessitate treatment as the first line therapy. An earlier diagnosis can lead to an invasive treatment if there’s no standard of care yet established, which most often would lead to invasive actions and thereby increase complications from overtreatment.
Frequency of testing. The optimal interval between screenings is also unclear: is it every 5 years, 1 year or 6 months?
Cost. It is unlikely that these tests will be covered by insurance and the out-of-pocket cost is likely to be around $950.
Dr. Susan Domchek of the Basser Center for BRCA at the University of Pennsylvania offers a compelling summary of the state of liquid biopsy screening tests
“We all want them to work…these tests are going to get better, but from what I’ve seen so far, these tests are not ready yet…Believe me, the minute we think these tests are good enough, we will roll them out to you!”
As a clinician I am excited and eager at the prospect of being able to offer such tests, and yet, the science is preliminary and not yet ready for prime time. The science is coming, the testing is underway, and “patience” is a necessary virtue…so let’s be patient and hopeful together.
In Real Health,
Dr. Ari Levy
Reference List
- Alix-Panabieres C. The future of liquid biopsy. Nature, 25 March2020. https://www.nature.com/articles/d41586-020-00844-5
- Liu MC, Oxnard GR, Klein EA, et al. Sensitive and specific multi-cancer detection and localization using methylation signatures in cell-free DNA. Ann Oncol. 2020; 31(6):745-759. https://pubmed.ncbi.nlm.nih.gov/33506766/
- Assessment of the Implementation of an Investigational Multi-Cancer Early Detection Test Into Clinical Practice. ClinicalTrials.gov. Study ID: NCT04241796. https://clinicaltrials.gov/ct2/show/NCT04241796?term=pathfinder%2C+grail&draw=2
- Nadauld LD, McDonnell CH, Beer TM, et al. The PATHFINDER Study: Assessment of the Implementation of an Investigational Multi-Cancer Early Detection Test into Clinical Practice. Cancers. 2021; 13(14):3501. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8304888/
- Kwo L, Aronson J. The Promise of Liquid Biopsies for Cancer Diagnosis. Evidence-Based Oncology. 2021; 27(7):SP261-SP262. https://www.ajmc.com/view/the-promise-of-liquid-biopsies-for-cancer-diagnosis
- Alba-Bernal A, Lavado-Valenzuela R, Dominguez-Recio ME, et al. Challenges and achievements of liquid biopsy technologies employed in early breast cancer. The Lancet. 2020; 62:102100. https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(20)30476-X/fulltext
- Xie L, Du X, Shi P, et al. Development and evaluation of cancer differentiation analysis technology: a novel biophysics-based cancer screening method. Expert Review of Molecular Diagnostics. 2022; 1:111-117. https://www.tandfonline.com/doi/full/10.1080/14737159.2021.2013201?src=recsys
- Srivastava S, Hanash S. Pan-Cancer Early Detection: Hype or Hope? Cancer Cell. 2020; 38(1):23-24. https://www.cell.com/cancer-cell/fulltext/S1535-6108(20)30271-3
- [image] Bardelli A, Pantel K. Liquid Biopsies, What We Do Not Know (Yet). Cancer Cell. 2017; 31(2):172-179. https://www.sciencedirect.com/science/article/pii/S1535610817300028
